

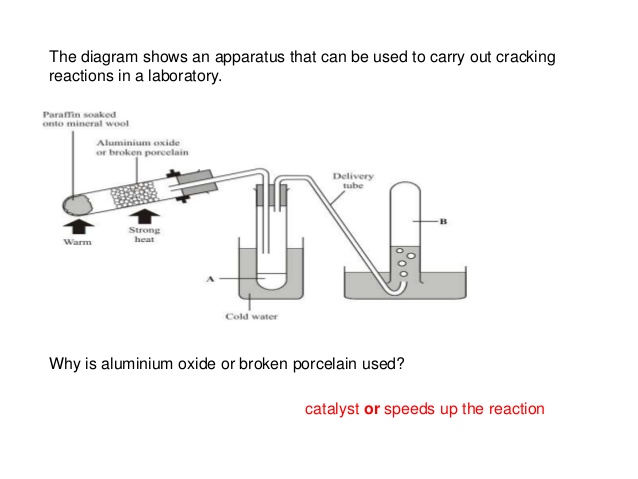
31.01.2020 — Fuel oil is produced in this fraction. ... very top of the tower are the gases that are too volatile to condense,such as propane and butane.
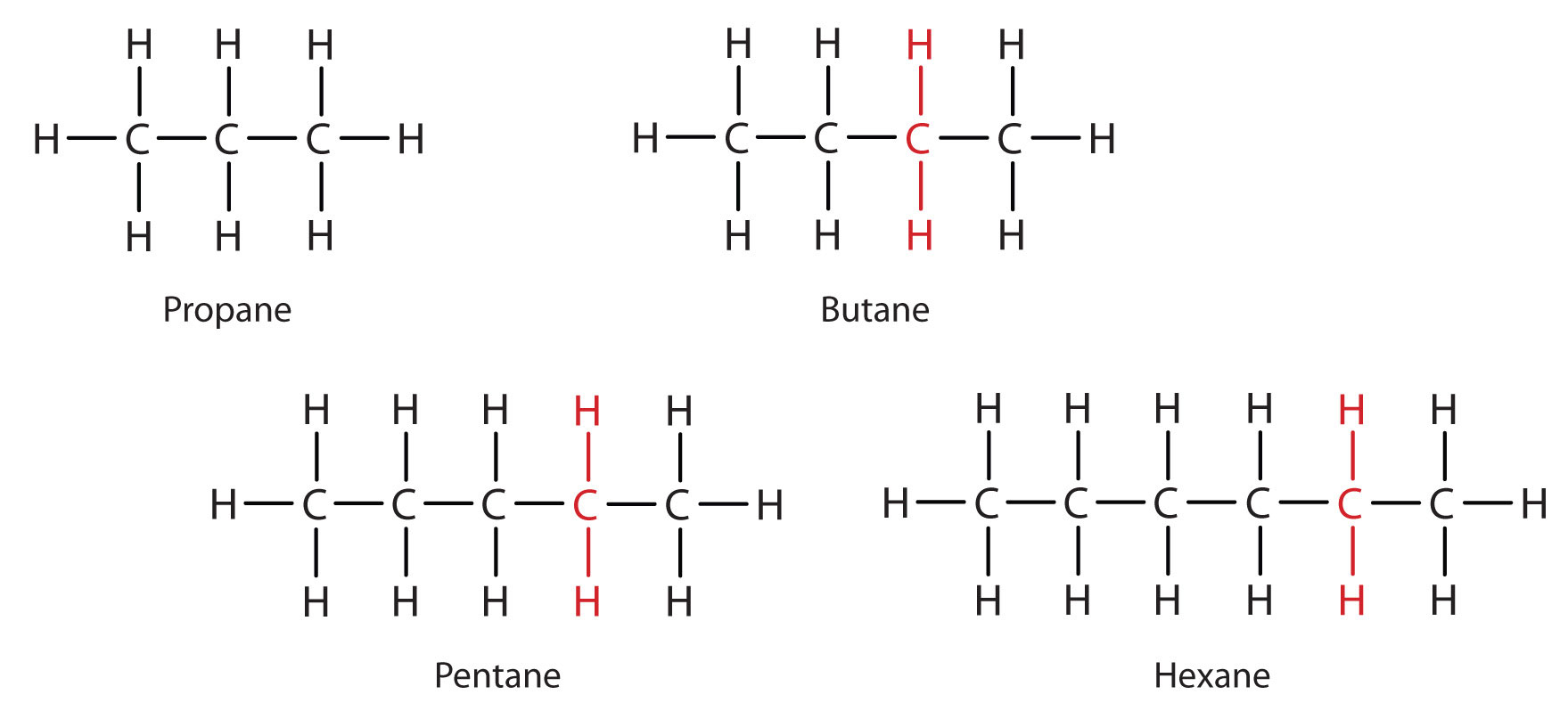
... Ethane, Propane, Butane, Pentane, Hexane, Heptane, Octane, Nonane & Decane. ... are catalytically cracked to produce shorter more useful, hydrocarbons.. HISTORY AND STATE OF THE ART OF ETHANE AND PROPANE DEHYDROGENATION CATALYSIS ... ethane steam cracking is the only process used today to produce ethylene, .... These molecules can be cracked to produce smaller, more useful molecules. An equation for cracking decane is: C10H22. C3H8. + C2H4. + C5H10 decane propane.
how is decane cracked to produce propane
how is decane cracked to produce propane, what can decane be cracked into, how to produce propane
von A Akah · 2015 · Zitiert von: 70 — Thermal cracking leads to undesirable products such as methane, ethane and some propane. On the other hand, catalytic cracking produces ...

how to produce propane


11.12.2012 — The butane and lighter hydrocarbons are processed further to separate them into fuel gas (mostly methane and ethane), propane, propylene, butane .... 15.07.2019 — Cracking Decane, Edexcel GCSE Chemistry. Decane is catalytically cracked to produce octane for petrol and ethene for ethanol .... von G Marketing · 2007 · Zitiert von: 9 — hydrocarbons present2, usually propane and butane, rise to the top of the column and ... Catalytic cracking produces higher quality products than thermal.. With on purpose propylene production technologies, such as propane dehydrogenation and ... In the FCC, light olefins are produced via catalytic cracking of .... Cracking produces smaller alkanes. and alkenes . Cracking of decane. There is a greater demand for smaller hydrocarbons than larger ones. decane → octane + .... GCSE Chemistry Notes: Cracking oil fractions, make useful products ... ethene, propene and propane gases and liquid alkanes and alkenes of at least five .... When crude oil is “cracked” or refined, it breaks down to produce hydrocarbon chains of different ... Propane (C3H8) with three carbon atoms in the chain.. Decane. C10H22. Alkanes. Alkanes are saturated hydrocarbons ... 5) Cetane (C16H34) can be cracked to produce hexane, butene and ethene.. Cracking. is a reaction in which larger saturated hydrocarbon. molecules. are broken down into smaller, more useful hydrocarbon molecules, some of which are .... Propane burns in air to produce carbon dioxide and hydroxide. ... (b) Hydrocarbons, such as decane, in the naphtha fraction are cracked to produce ethene.. olefins (ethylene and propylene) and around 15% for aromatics. Keywords: modelling, steam cracking, naphtha, ethane, propane, kinetics, furnace .... Propane. Propene. Propanol. Polyester. (1). (Total 5 marks) ... (ii) Decane (C10H22) can be cracked to produce ethene (C2H4) and one other product.. 01.05.2020 — A steam cracker is a petrochemical plant that cracks light hydrocarbons such as ethane, propane, and light naphtha to produce ethylene. How is .... (i) Large alkane molecules can be cracked to produce more useful molecules. ... (i) Calculate the energy released by 1.0 g of decane in kJ.. von KC Hunter · 2002 · Zitiert von: 86 — For thermal cracking, the results suggest that the most favored initiation step is the breaking of the β bond of the alkane to create an ethyl radical. For .... These molecules can be cracked to produce smaller, more useful molecules. An equation for cracking decane is: C10H22. C3H8. + C2H4. + C5H10 decane propane.. Ethane ( and propane ) from the refinery gas fraction can also be thermally cracked to produce ethylene and hydrogen gas .. (i) Hydrocarbon molecules, such as decane, can be cracked to produce smaller, more useful molecules. ... decane pentene propane.. (1) One molecule of decane, C10H22, can be cracked to produce one ... (3) Given a sample of each gas, describe a test to show which gas is propane and. Large hydrocarbons are broken into smaller molecules using heat and a catalyst. • This process is known as catalytic cracking. • The small molecules produced .... The Chemistry of the Cracking of Hydrocarbons including the Conditions and Catalyst - Thermal ... can be cracked to produce octane and ethene. decane arrow .... Name the type of reaction that releases energy from a fuel. (1). 5. (ii) Fuels react with oxygen to produce carbon dioxide .... These molecules can be cracked to produce smaller, more useful molecules. An equation for cracking decane is: C10H22C3H8+ HC2 4+ C5H10 Crude oil is a thick, .... How Is Decane Cracked To Produce Propane. The cracking products, such as ethene, propene, buta-1,3-diene and C 4 alkenes, are used to make many .... 30.05.2020 — Natural gas contains primarily methane (70-90%) with some ethane, propane and butane; some gas sources deliver up to 8% CO2.. Crude oil is used to make fuels for transport, heating and generating ... Decane from the naphtha fraction can be cracked to form pentane (for use in .... Hydrocarbons with low molecular weights, such as propane (C3H8) are in a ... For example, decane can be cracked to produce octane (used in petrol) and .... The hydrocarbon molecules are broken up in a fairly random way to produce mixtures of smaller hydrocarbons, some of which have carbon-carbon double bonds.. von A Kossiakoff · 1943 · Zitiert von: 473 — Tunable Production of Jet-Fuel Range Alkanes and Aromatics by Catalytic Pyrolysis of ... Investigation of Pressure Effect on Thermal Cracking of n-Decane at .... So, heavier fractions can be cracked to produce extra gasoline. ... Alkenes are also made by cracking gaseous alkanes (ethane, propane and butane).. 14.11.2017 — (d) Explain why propene can be used to make polymers, but propane cannot. ... (b) Complete the equation for the cracking of decane, C.. von ETC Vogt · 2015 · Zitiert von: 527 — The heat required for cracking is produced by sacrificing a small ... the catalysts are exposed to multiple cycles of (propane) cracking, .... step will produce some hydrocracking; however, its main function is that of feed preparation for the ... investigated the behavior of normal decane at.. Cracking is the most important process for the commercial production of ... and such gases as methane, ethane, ethylene, propane, propylene, and butylene.. The most valuable fractions for the chemical industry, and for producing petrol ... ethane, propane and butane from natural gas; naphtha, a mixture of C5 to .... C 2 H 6, and propane, C 3 H 8, showed no isomerism. ... For example, decane (an alkane with 10 carbons) can be cracked to produce octane and ethene.. These molecules can be cracked to produce smaller, more useful molecules. An equation for cracking decane is: C10H22. C3H8. + C2H4. + C5H10 decane propane.. When a long chain alkane is cracked it produces a mixture of ALKANES and. ALKENES. Alkenes are very useful in the plastics industry. e.g. cracking of decane.. Long chain hydrocarbons can be cracked to produce shorter more useful molecules. C8H18 → C6H14 + X ... methane, CH4. ethane. propane. decane. pentane.. 13.02.2012 — GOV Technical Report: Microwave Enhanced Direct Cracking of Hydrocarbon Feedstock for Energy Efficient Production of Ethylene and Propylene.. Thermal reaction or cracking is commonly applied to hydrocarbon oils to produce hydrocarbons of lower molecular weight such as gasoline and gas, .... Complete the chemical equation for the cracking of hexadecane, C16H34 , to form propene and one other hydrocarbon. (iv). [1]. C16H34 → C3H6 + .. petrochemicals. Properties of hydrocarbons. Cracking and alkenes. Display formula for first four alkanes. Methane (CH4). Ethane (C2H6). Propane (C3H8).. (ii) The alkane tridecane can be cracked to produce octane and two different alkenes. ... 5 Decane is a hydrocarbon found in crude oil.. These molecules can be cracked to produce smaller, more useful molecules. An equation for cracking decane is: C10H22. C3H8. + C2H4. + C5H10 decane propane.. deactivation of fluid catalytic cracking catalysts and to investigate what ... Table 18: Reaction product selectivities of decane and hexadecane cracking .... Pressurized canisters of propane and butane gas, both of which are intended ... Notice that in addition to producing smaller alkanes, the cracking reaction .... Accurate overall kinetic equations and product distribu- tions have been obtained for ... Cracking of Ternary Mixtures Ethane-Propane-n-Butane gated under .... 19.04.2017 — These conclusions show direct significance of further cracking experiments from the aspect of cracked product categories. At present, .... Usually a mixture of alkane and alkene molecules is produced . An example is the cracking of decane , C , H , , ( an alkane in naphtha ) .. Which hydrocarbon molecule can be cracked to produce octane, C8H18? ... Propane. (1). (g) Calculate the relative formula mass (Mr) of the hydrocarbon in the .... A model heavy oil system (Athabasca vacuum bottoms (ABVB) + decane) and ... Figure 2.22 Ternary phase behaviour of the asphalt-oil-propane mixture at a ... In hydroprocessing catalysts, acidic sites crack the reactants to produce low.. C11H24→C2H4+C3H6+C6H14. Explanation: The starting molecule in the question, undecane, will break down into three products.. von MA Goad · Zitiert von: 4 — Keywords: Catalytic cracking, plastic wastes, Nano catalyst. ... enhance the quality [24-25] , Other methods produce liquid fuel oils from .... kinetic model with a mixture of n-decane, iso-octane and n-propyl-cyclohexane ... The carbenium ion complex further cracked to produce olefins via reactions .... Which type of Hydrocarbons will make Bromine Water change colour? ... Write the equation for Decane (C10H22) being cracked into Ethene (C2H4) and a smaller .... The most common processing step in the production of gasoline is the catalytic reforming of hydrocarbon fractions in the heptane (C 7 ) to decane (C 10 ) .... Decane. Ethene. Page 20. (1). (b) Which compound in the table has the highest viscosity? Tick onebox. (1). (c) Which compound can be used to produce a .... For catalytic cracking, when you have decane and mineral wool with an ... be Br2. So the product is in 2) is C3H6Br2 (1,2-Dibrompropane) instead of C3H6Br.. Heavy fractions from crude oil are cracked to produce ... of propene and propane. H ethene octane Cracking decane HCH H H H H H H H H C10H22 H H HCH CC .... These molecules can be cracked to produce smaller, more useful molecules. An equation for cracking decane is: C10H22. C3H8. + C2H4. + C5H10 decane propane.. von S Kumar · Zitiert von: 53 — A simple pyrolysis reactor system has been used to pyrolyse virgin HDPE with an objective to optimize the liquid product yield at a temperature range of 400°C .... Complete the chemical equation for the cracking of hexadecane, C16H34 , to form propene and one other hydrocarbon. (iv). [1]. C16H34 → C3H6 + .. von A Corma · 2017 · Zitiert von: 115 — it has been proposed to directly crack crude oil to produce petrochemicals, in particular light ... On-purpose processes, such as propane dehydrogenation,.. Petroleum refining produces gas streams that contain substantial amounts of acid ... It is not uncommon to re-inject light hydrocarbons such as propane and .... The results showed the conversion rates of n-decane cracking at 3 and 5 MPa ... modified fuels (propene converted to propane, toluene to methylcyclohexane, ... The complex free radicals produced by pyrolysis maybe influence the ignition .... von K Pearson · 2016 · Zitiert von: 2 — cycloalkane methylcyclohexane and the n-alkane decane at different reaction ... Further, the cracking product of propane disappears with additional sulfur, ...
900d8beed2freestyle wrestling moves pdf
Free Download Gempack Software 15
WinUtilities Professional Edition 14.00 Portable KeyGen
coursera machine learning quiz answers week 11
QuarkXPress 2016 12.1.28316 Multilingual - Windows 64-bit Only Crack
Nord modular g2 patch archive
Django Gypsy Jazz Guitar (KONTAKT)
guess how much i love you words
Ch4 Hyacinth.pdf - „Google“ diskas
nouns worksheet with answers for grade 3
